PRODUCTS
NGS-based liquid biopsy is a rapidly evolving application that requires accurate and precise reference standards. The Twist Pan-cancer Reference Standard is a high-quality, standardized control for use in the research and development of NGS-based liquid biopsy assays. This reference standard is ideal for establishing the analytical limit of detection (LoD) for specific cancer variants and as a control to track the quality of an NGS assay workflow and assess the fidelity of the assay process during research and development of such assays.
458 unique naturally occurring cancer variants
132 clinically relevant variants
Covers 84 different genes involved in cancer
Background DNA derived from human cfDNA samples
DNA size profile and post-sequencing profile mimic native cfDNA
Seven individual VAF percentages to choose from
Digital Droplet PCR verification of VAF percentages
Convenient test set of all VAF percentages available
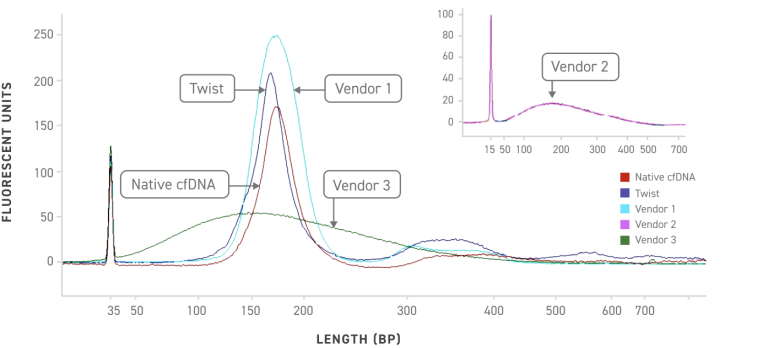
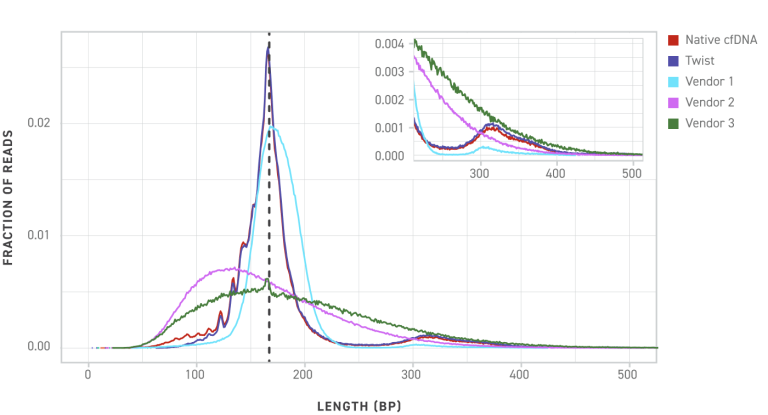
DNA analysis of the Twist cfDNA Pan-Cancer Reference Standard and competitors reference standards show that Twist more closely mimics native cell-free DNA providing a more true to life test result.
Post sequencing library size distribution comparison between Twist and competitors show how similar Twist mimics native cfDNA samples including the jagged leading peaks.
Get a call from your local Decode Science representative to help you find the best fit genomics products for you.
Or give us a call at:
This site uses cookies. By continuing to browse the site, you are agreeing to our use of cookies.
OKLearn moreWe may request cookies to be set on your device. We use cookies to let us know when you visit our websites, how you interact with us, to enrich your user experience, and to customize your relationship with our website.
Click on the different category headings to find out more. You can also change some of your preferences. Note that blocking some types of cookies may impact your experience on our websites and the services we are able to offer.
These cookies are strictly necessary to provide you with services available through our website and to use some of its features.
Because these cookies are strictly necessary to deliver the website, refusing them will have impact how our site functions. You always can block or delete cookies by changing your browser settings and force blocking all cookies on this website. But this will always prompt you to accept/refuse cookies when revisiting our site.
We fully respect if you want to refuse cookies but to avoid asking you again and again kindly allow us to store a cookie for that. You are free to opt out any time or opt in for other cookies to get a better experience. If you refuse cookies we will remove all set cookies in our domain.
We provide you with a list of stored cookies on your computer in our domain so you can check what we stored. Due to security reasons we are not able to show or modify cookies from other domains. You can check these in your browser security settings.
These cookies collect information that is used either in aggregate form to help us understand how our website is being used or how effective our marketing campaigns are, or to help us customize our website and application for you in order to enhance your experience.
If you do not want that we track your visit to our site you can disable tracking in your browser here:
We also use different external services like Google Webfonts, Google Maps, and external Video providers. Since these providers may collect personal data like your IP address we allow you to block them here. Please be aware that this might heavily reduce the functionality and appearance of our site. Changes will take effect once you reload the page.
Google Webfont Settings:
Google Map Settings:
Google reCaptcha Settings:
Vimeo and Youtube video embeds:
The following cookies are also needed - You can choose if you want to allow them:
Simply add your details below and get quick access to the brochure.
Simply add your details below and get quick access to the brochure.
Simply add your details below and get quick access to the brochure.
